Rilemo's device is a portable, non-ionising brain imaging system designed to bring rapid, point-of-care neurological imaging to the bedside, the emergency room, the intensive care unit, and (in the next phase of expansion) the ambulance and the pre-hospital setting. It produces clinically interpretable images in seconds, on any patient, in almost any environment, and is designed to complement conventional imaging modalities such as CT and MRI rather than replace them.

This page describes what the device is and how it is used. The underlying technology and physics are presented in Technology Overview . The imaging evidence is in Clinical Validation. The clinical and economic context is in Market Overview. The commercial deployment plan is in Go-to-Market.
The System
Rilemo's device is built as a three-component medical system, supplemented by a single-use sterile consumable.
Component | Role | Where it sits during use |
Head Unit | The wearable imaging device, containing the antennas, the front-end electronics, and the on-board sensors. | On the patient’s head, secured with a soft pressure comparable to a sweatband. |
Bedside Unit | The compute and control station, hosting the AI image-reconstruction pipeline, the encrypted storage, the colour touchscreen, and the wireless link to the Head Unit. | Next to the patient, on a cart, table, or trolley. |
Calibration Phantom | A small portable cylinder used during the start-up calibration cycle. | Stored next to the Bedside Unit, used at start-up and on demand. |
Single-use head cover (consumable) | A sterile, medical-grade barrier film placed between the patient’s head and the Head Unit, replaced for every patient. | Stored in an accessible pocket on the Bedside Unit. |
The underlying physics of the imaging method, and the defensibility of the system architecture are presented in the Technology Overview section.
How a scan is performed
A complete scan, from the moment the device is taken out of standby to the moment the result is shown to the doctor, follows four steps and takes under 2 minutes in total.
No patient preparation is required. The patient does not need to undress, remove jewellery, or have their hair handled. There is no shielded room, no contrast injection, no closed-bore environment, and no operator PPE. The patient can be lying in a hospital bed, sitting in a triage chair, or strapped on a stretcher.
1. Head cover and Head Unit placement. The clinician takes a sterile single-use head cover from the Bedside Unit, places it on the patient's head, and secures the Head Unit with a soft pressure comparable to a sweatband. The fit accommodates the full target patient population from the 5th to the 95th percentile head size, validated across a large set of anatomically realistic head models through electromagnetic digital twin simulations.
2. Boot and calibration in under 60 seconds. From device-off to ready for imaging, the system completes its boot and calibration cycle in less than 60 seconds.
3. Image acquisition and result display in under 5 seconds. The clinician triggers a scan from the Bedside Unit. The acquisition takes less than 5 seconds. Within seconds, the AI reconstruction pipeline produces a three-modality image set on the Bedside Unit colour display.
The entire sequence is executable by any trained clinician, emergency physicians, neurologists, neurosurgeons, and intensive care staff, without requiring radiological expertise, directly addressing the structural gap in bedside imaging identified across Rilemo's structured clinician interviews, summarised in Market Overview.
Two operational modes
The same hardware supports two clinically distinct usage modes.
Single-image mode. The Head Unit operates on its own internal battery and communicates with the Bedside Unit over an encrypted wireless link. No cable to the patient. One scan is acquired, reconstructed, and exported. This is the mode used for triage in the emergency room and first assessment in pre-hospital settings, where one device can serve multiple patients in sequence.
Monitoring mode. The Head Unit is cable-connected to the Bedside Unit and produces repeated scans at configurable time intervals. This is the mode used for serial imaging of unstable neurological patients in intensive care, in the post-operative window, and during therapy monitoring, where the clinical value lies in tracking the evolution of a patient's neurological status across multiple timestamps over time. In this mode, one device is dedicated to one patient for the duration of the imaging session.
The two modes reflect distinct clinical workflows that can coexist within the same department.
What the product produces
For every scan, the device produces a three-modality image set:
- Tomography: anatomical reconstruction of the imaged region.
- Radar (contrast) image: structural transitions and boundaries inside the imaged volume.
- Dielectric Permittivity map: quantitative distribution of fluid-related tissue properties.

The visual reference of what the three-modality output looks like on a healthy head is presented in Technology Overview. The diagnostic discrimination between healthy and pathological cases (central haemorrhagic events, wedge haemorrhagic strokes, and the cadaver experimental campaign) is presented in Clinical Validation.
The output is DICOM-compatible (the standard medical imaging format) and can be exported to the hospital PACS (Picture Archiving and Communication System), kept locally on the Bedside Unit's encrypted storage, or fused with existing CT, MRI, or X-ray images for joint interpretation. The fusion approach allows clinicians to read the device's output directly on top of imaging they already know, overlaying fluid-related functional information on a familiar anatomical reference.
First indication: acute neurological conditions
The first commercial use case is acute neurological conditions, where the value of bringing imaging to the patient is at its highest. The conditions being designed and validated against in this first phase are:
Acute stroke (haemorrhagic and ischaemic),
where rapid identification and localisation of the affected region directly supports time-sensitive treatment decisions.
Traumatic brain injury and head trauma,
where bedside imaging supports early triage between conservative management, further imaging, and immediate surgical intervention.
Intracranial bleeding and post-surgical complications,
where repeated point-of-care imaging supports identification and monitoring of evolving haemorrhagic events that today require patient transport to centralised radiology.
The validation work supporting these clinical targets, including the in-silico digital-twin campaign on healthy and pathological head models, the cadaver experiments at clinically relevant haemorrhagic volumes, and the broader validation portfolio, is presented in Clinical Validation.
Future clinical applications
While neurological conditions represent the first commercial entry point, the underlying technology is applicable to other clinical domains where imaging of fluid-related tissue properties creates clinical value.
- Respiratory conditions - pleural effusions, pulmonary oedema, and inflammatory processes.
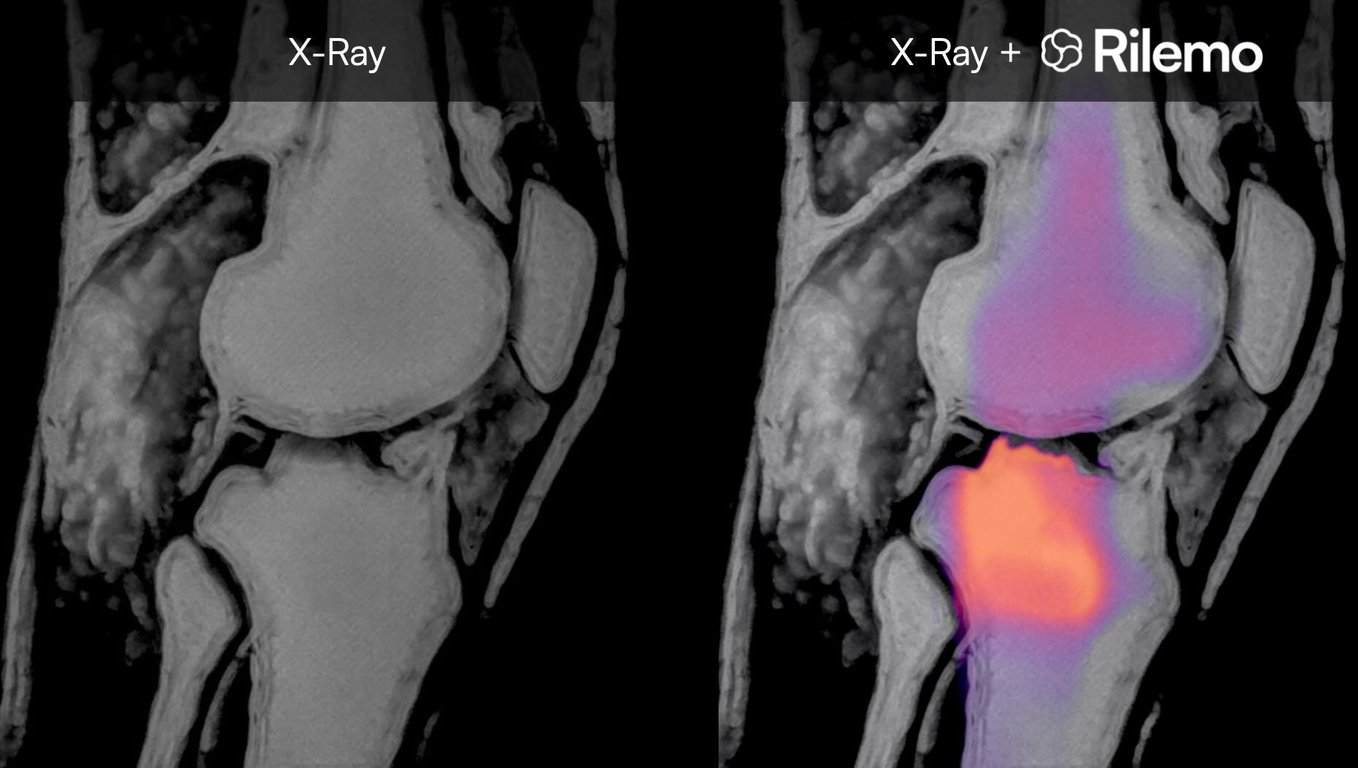
- Orthopaedic pathologies - joint effusions, inflammatory conditions, and post-traumatic fluid accumulation.
- Breast screening - fluid-related abnormalities in environments where conventional screening infrastructure is unavailable.
- Field surgery support - real-time imaging aid during surgical interventions in field hospitals or low-resource settings.
These adjacencies represent a significant expansion of the addressable market beyond the current scope. Each can be pursued without a fundamental redesign of the device, leveraging the same core technology across new indications and regulatory cycles as the company matures along the path.
This Data Room is provided to you by:

Rilemo S.r.l.
Via Don Sebastiano Colleoni 20, Robbiate (LC), 23899, Italy
EU VAT: IT 04187140134 - ATECO: 26.60.02
PEC: - rilemo@legalmail.it
